| Référence associée : | | |
| Long versus short course treatment with metformin and clomiphene citrate for ovulation induction in women with PCOS .. cacher .... voir plus ..
Background Background Polycystic ovary syndrome (PCOS) is the most common endocrinopathy among reproductive-aged women. Apart from infertility, women with PCOS often have other endocrine disorders, including insulin resistance, hyperinsulinaemia and hyperandrogenism. Metformin,combined with clomiphene citrate (CC), has been shown to be more effective in ovulation induction when compared with clomiphene citrate alone. The optimal duration for metformin pretreatment before initiation of clomiphene citrate, however, is unknown. Objectives Objectives To determine the effectiveness of short-course (less than four weeks) metformin plus CC versus long-course (four weeks or more) metformin plus CC with regard to ovulation and achievement of pregnancy in infertile women with PCOS. Search methods Search methods We searched the Cochrane Menstrual Disorders and Subfertility Group Trials Register, the Cochrane Central Register of Controlled Trials, MEDLINE, CINAHL, EMBASE and PsycINFO (all from inception to 1 February 2012). Selection criteria Selection criteria Randomised controlled trials comparing short-course (less than four weeks) metformin plus CC versus long-course (four weeks or more) metformin plus CC for ovulation or achievement of pregnancy in infertile women with PCOS. Data collection and analysis Data collection and analysis No trials were found that met the selection criteria. Main results Main results No randomised controlled trials were identified. Authors conclusions Authors conclusions There are insufficient data to determine whether short-course metformin pretreatment is as effective as the conventional long-course metformin pretreatment before initiation of clomiphene citrate for ovulation induction in infertile women with PCOS. A well-designed randomised controlled trial is needed to answer this important clinical question.
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Blood flow changes in the ovarian and uterine arteries in women with polycystic ovary syndrome who respond to clomiphene citrate: correlation with serum hormone concentrations .. cacher .... voir plus ..
Objective Our purpose was to study the hemodynamic changes in the uterine and intraovarian vessels in women with polycystic ovary syndrome (PCOS) and to relate the vascular changes to hormonal indices. Methods Six women with a history of previous anovulatory cycles and/or oligomenorrhea and/or elevated serum luteinizing hormone concentrations in addition to polycystic ovaries underwent induction of ovulation with clomiphene citrate. Serial transvaginal ultrasonography was performed on menstrual cycle day 2, daily from the estimated day of ovulation minus 6, 6-hourly from the estimated day of ovulation minus 2 or when the mean follicular diameter was greater than 16 mm (whichever was earlier) until 6 h after follicular rupture, and once 7 days after follicular rupture. At each scan, uterine and intraovarian blood flow was assessed by transvaginal color Doppler ultrasound, and serum concentrations of follicle stimulating hormone (FSH), luteinizing hormone (LH), estradiol and progesterone were assessed by fluoroimmunoassay. Results All six women demonstrated ultrasonographic features of ovulation. Follicular peak systolic blood flow velocity (Vmax) and ovarian stromal Vmax in the dominant ovary increased during the follicular phase of the menstrual cycle and reached a peak at 36 and 42 h after the onset of the LH surge of 30.7 ± 4.6 cm/s and 37.0 ± 3.9 cm/s, respectively. There was no significant change in the ovarian stromal Vmax of the non-dominant ovary. There was no significant change in the pulsatility index (PI) in either the follicular or the ovarian stromal vessels during the menstrual cycle. The changes in follicular Vmax correlated significantly with changes in serum concentrations of FSH, LH and progesterone. Uterine artery time-averaged maximum velocity (TAMX) declined to its lowest levels at LH surge minus 6 days, and thereafter increased. In contrast, the PI increased during the follicular phase of the cycle, reaching a peak at LH surge minus 4 days, and thereafter declined. The changes in uterine artery PI correlated inversely with serum estradiol and serum progesterone concentrations. Conclusions Women with PCOS have significant differences in intraovarian and uterine artery hemodynamics, both at the baseline ultrasound scan and during the clomiphene-induced menstrual cycle, compared with women with normal ovaries. The intraovarian and uterine artery vascular differences are likely to be due to a primary disorder within the polycystic ovary and their different hormonal status. Clomiphene citrate may have an effect on uterine artery blood flow. Copyright © 1998 International Society of Ultrasound in Obstetrics and Gynecology
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Le SPOK est un des cas où la majorité des femmes reçoivent un traitement inadéquat malgré les risques à long terme qu'elles encourent.
Legro RS, Urbanek M, Kunselman AR, et al: Self-Selected Women with Poly cys tic Ovary Syndrome are
Re pro duc tive ly and Metabolically Abnormal and Under Treated. Fertil Steril 78:51-57, 2002. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| The management of infertility associated with polycystic ovary syndrome .. cacher .... voir plus ..
Polycystic ovary syndrome [PCOS] is the commonest cause of anovulatory infertility. Treatment modes available are numerous mainly relying on ovarian stimulation with FSH, a reduction in insulin concentrations and a decrease in LH levels as the basis of the therapeutic principles. Clomiphene citrate is still the first line treatment and if unsuccessful is usually followed by direct FSH stimulation. This should be given in a low dose protocol, essential to avoid the otherwise prevalent complications of ovarian hyperstimulation syndrome and multiple pregnancies. The addition of a GnRH agonists, while very useful during IVF/ET, adds little to ovulation induction success whereas the position of GnRH antagonists is not yet clear. Hyperinsulinemia is the commonest contributor to the state of anovulation and its reduction, by weight loss or insulin sensitizing agents such as metformin, will alone often restore ovulation or will improve results when used in combination with other agents. Laparoscopic ovarian drilling is proving equally as successful as FSH for the induction of ovulation, particularly in thin patients with high LH concentrations. Aromatase inhibitors are presently being examined and may replace clomiphene in the future. When all else has failed, IVF/ET produces excellent results. In conclusion, there are very few women suffering from anovulatory infertility associated with PCOS who cannot be successfully treated today.
... lire plus sur ce sujet dans un nouvel onglet.... | | |  |
| Des ovaires polykystiques ont été mis en évidence par échographie chez 87% des patientes avec des cycles irréguliers et 92% présentant un hirsutisme idiopathique.
Adams J, Polson DW, Franks S: Prevalence of Polycystic ovaries in Women with Ano vu la tion and Idiopathic
Hir sut ism. Br Med J 293:355-359, 1986 .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Il a été observé un taux d'incidence de 14.2% du SOPK dans le groupe de femmes asymptomatiques, l'incidence était plus important dans le groupe des femmes symptomatiques.
L'insulino-résistance est présente chez plus de 50% des femmes présentant un SOPK.
Koivunen R, Laatikainen T, Tomas C, et al: The Prev a lence of Polycystic Ovaries in Healthy Women. Acta
Obstet Gynecol Scand 78:137-141, 1999 .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| [Using an ovarian drilling by hydrolaparoscopy or recombinant follicle stimulating hormone plus metformin to treat polycystic ovary syndrome: Why a randomized controlled trial fail?] .. cacher .... voir plus ..
OBJECTIVES: To evaluate pregnancy rates after randomized controlled trial (RCT) between ovarian drilling by fertiloscopy or ovarian hyperstimulation+insemination+metformine after clomifène citrate (cc) treatment fails. PATIENTS AND METHODS: Randomized controlled trial with 126 patients in each arm in 9 university centers. After 6-9 months of stimulation by cc, 2 groups were randomized: group 1, ovarian drilling with bipolar energy versus group 2: 3 months treatment by metformine followed by 3 hyperstimulation by FSH+insemination. The success rate was pregnancy rate above 12 weeks. RESULTS: RCT was stopped after the screening of 40 patients. In spite of the low number of patients, the pregnancy rate is significantly higher in medical group 8/16 versus 3/18 (p=0.04). CONCLUSION: The causes of fail of RCT were in relationship with difficulties of inclusion, with absence of final agreement by team included. Moreover, RCT between medical and surgical management is often root of difficulties for patients who decline surgical strategy. However, medical treatment appeared better than drilling in this RCT.
... lire plus sur ce sujet dans un nouvel onglet.... | | |  |
| Stein IF, Leventhal ML: Amenorrhea Associated with Bi lat er al Polycystic Ovaries. Am J Obstet Gynecol Page
181-191, 1934. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Ovarian-adrenal cross-talk in polycystic ovary syndrome: evidence from wedge resection .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | | 
|
| Myoinositol as a Safe and Alternative Approach in the Treatment of Infertile PCOS Women: A German Observational Study .. cacher .... voir plus ..
The use of 2 × 2000 mg myoinositol + 2 × 200 μg folic acid per day is a safe and promising tool in the effective improvement of symptoms and infertility for patients with a polycystic ovary syndrome (PCOS). Using a questionnaire an observational study was performed under German gynecologists to collect data on ovulation and pregnancy rates in PCOS patients with infertility. In this observational study, 3602 infertile women used myoinositol and folic acid between 2 and 3 months in a dosage of 2 × 2000 mg myoinositol + 2 × 200 μg folic acid per day. In a subgroup of 32 patients, hormonal values for testosterone, free testosterone, and progesterone were analyzed before and after 12 weeks of treatment. The mean time of use was 10.2 weeks. During this time 70% of these women had a restored ovulation, and 545 pregnancies were obtained. This means a pregnancy rate of 15.1% of all the myoinositol and folic acid users. In 19 cases a concomitant medication with clomiphene or dexamethasone was used. One twin pregnancy was documented. Testosterone levels changed from 96.6 ng/ml to 43.3 ng/ml and progesterone from 2.1 ng/ml to 12.3 ng/ml (p < 0.05) after 12 weeks of treatment. No relevant side effects were present among the patients. This study could show that a new treatment option for patients with a PCOS and infertility is available. The achieved pregnancy rates are at least in an equivalent or even superior range than those reported by the use of metformin.
... lire plus sur ce sujet dans un nouvel onglet.... | | | 
|
| Koivunen R, Laatikainen T, Tomas C, et al: The Prev a lence of Polycystic Ovaries in Healthy Women. Acta
Obstet Gynecol Scand 78:137-141, 1999. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Clomiphene and other antioestrogens for ovulation induction in polycystic ovarian syndrome .. cacher .... voir plus ..
Background Background Subfertility due to anovulation is a common problem in women. First-line oral treatment is with antioestrogens such as clomiphene citrate, but resistance may be apparent with clomiphene. Alternative and adjunctive treatments have been used including tamoxifen, dexamethasone, and bromocriptine. The effectiveness of these is to be determined. Objectives Objectives To determine the relative effectiveness of antioestrogen agents including clomiphene alone or in combination with other medical therapies in women with subfertility associated with anovulation, possibly caused by polycystic ovarian syndrome. Search methods Search methods We conducted a search of the Cochrane Gynaecology and Fertility Group Trials Register, the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, Embase, PsycINFO, and CINAHL (all from inception to August 2016) to identify relevant randomised controlled trials (RCTs). We searched the United Kingdom National Institute for Clinical Excellence (NICE) guidelines and the references of relevant reviews and RCTs. We also searched the clinical trial registries for ongoing trials (inception until August 2016). Selection criteria Selection criteria We considered RCTs comparing oral antioestrogen agents for ovulation induction (alone or in conjunction with medical therapies) in anovulatory subfertility. We excluded insulin-sensitising agents, aromatase inhibitors, and hyperprolactinaemic infertility. Data collection and analysis Data collection and analysis Two review authors independently performed data extraction and quality assessment. The primary outcome was live birth; secondary outcomes were pregnancy, ovulation, miscarriage, multiple pregnancy, ovarian hyperstimulation syndrome, and adverse effects. Main results Main results This is a substantive update of a previous review. We identified an additional 13 studies in the 2016 update. The review now includes 28 RCTs (3377 women) and five RCTs awaiting classification. Five of the 28 included trials reported live birth/ongoing pregnancy. Secondary outcomes were poorly reported. The quality of the evidence ranged from low to very low. The primary reasons for downgrading the evidence were imprecision and risk of bias associated with poor reporting. Antioestrogen versus placebo Live birth rate, miscarriage rate, multiple pregnancy rate, and ovarian hyperstimulation syndrome (OHSS) No data were reported for these outcomes. Clinical pregnancy rate Clomiphene citrate was associated with an increased chance of a clinical pregnancy compared with placebo, though the size of the benefit was very uncertain (odds ratio (OR) 5.91, 95% confidence interval (CI) 1.77 to 19.68; 3 studies; 133 women; low-quality evidence). If the chance of a clinical pregnancy was 5% in the placebo group, then between 8% and 50% of women would have a clinical pregnancy in the clomiphene group. Clomiphene citrate versus tamoxifen Live birth rate There was no clear evidence of a difference in the chance of a live birth between the clomiphene citrate and tamoxifen groups (OR 1.24, 95% CI 0.59 to 2.62; 2 studies; 195 women; low-quality evidence). If 20% of women in the tamoxifen group had a live birth, then between 13% and 40% of women in the clomiphene citrate group would have a live birth. Miscarriage rate There was no clear evidence of a difference in the chance of a miscarriage between the clomiphene citrate and tamoxifen groups (OR 1.81, 95% CI 0.80 to 4.12; 4 studies; 653 women; low-quality evidence). If 3% of women in the tamoxifen group had a miscarriage, then between 2% and 10% in the clomiphene citrate group would have a miscarriage. Clinical pregnancy rate There was no clear evidence of a difference in the chance of a clinical pregnancy between the clomiphene citrate and tamoxifen groups (OR 1.30, 95% CI 0.92 to 1.85; 5 studies; 757 women; I2 = 69%; low-quality evidence). If 22% of women in the tamoxifen group had a clinical pregnancy, then between 21% and 35% in the clomiphene citrate group would have a clinical pregnancy. Multiple pregnancy rate There was insufficient evidence of a difference in the chance of a multiple pregnancy between the clomiphene citrate group (OR 2.34, 95% CI 0.34 to 16.04; 3 studies; 567 women; very low-quality evidence). If 0% of women in the tamoxifen group had a multiple pregnancy, then between 0% and 0.5% of women in the clomiphene group would have a multiple pregnancy. OHSS There were no instances of OHSS in either the clomiphene citrate or the tamoxifen group reported from three studies. Clomiphene citrate with tamoxifen versus tamoxifen alone Clinical pregnancy rate There was insufficient evidence to determine whether there was a difference between groups (OR 3.32, 95% CI 0.12 to 91.60; 1 study; 20 women; very low-quality evidence). No data were reported for the other outcomes. Other comparisons of interest Limited evidence suggested that compared with a gonadotropin, clomiphene citrate was associated with a reduced chance of a pregnancy, ongoing pregnancy, or live birth, with no clear evidence of a difference in multiple pregnancy rates. The comparison of clomiphene citrate plus medical adjunct versus clomiphene alone was limited by the number of trials reporting the comparison and poor reporting of clinical outcomes relevant to this systematic review and by the number of adjuncts reported (ketoconazole, bromocriptine, dexamethasone, combined oral contraceptive, human chorionic gonadotropin, hormone supplementation). The addition of dexamethasone or combined oral contraceptive suggested a possible benefit in pregnancy outcomes, but findings were very uncertain and further research is required to confirm this. There was limited evidence suggesting that a 10-day regimen of clomiphene citrate improves pregnancy outcomes compared with a 5-day regimen. Data for early versus late regimens of clomiphene citrate were insufficient to be able to make a judgement on differences for pregnancy outcomes. Authors conclusions Authors conclusions We found evidence suggesting that clomiphene citrate improves the chance of a clinical pregnancy compared with placebo, but may reduce the chance of live birth or ongoing pregnancy when compared with a gonadotropin. Due to low event rates, we advise caution interpreting these data. The comparison of clomiphene citrate plus medical adjunctive versus clomiphene alone was limited by the number of trials reporting the comparison. The evidence was very low quality and no firm conclusions could be drawn, but very limited evidence suggested a benefit from adjunctive dexamethasone or combined oral contraceptives. Low-quality evidence suggested that a 10-day regimen of clomiphene citrate improves pregnancy rates compared with a 5-day regimen, but further research is required.
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Adams J, Polson DW, Franks S: Prevalence of Polycystic ovaries in Women with Ano vu la tion and Idiopathic
Hir sut ism. Br Med J 293:355-359, 1986. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Correlation of Endometrial Glycodelin Expression and Pregnancy Outcome in Cases with Polycystic Ovary Syndrome Treated with Clomiphene Citrate Plus Metformin: A Controlled Study .. cacher .... voir plus ..
Objective. The purpose of this study was to evaluate the relationship between clomiphene citrate (CC) plus metformin treatment and endometrial glycodelin expression and to then correlate this relationship with pregnancy outcomes. Material and Methods. A total of 30 patients diagnosed with polycystic ovary syndrome (PCOS) according to the Rotterdam criteria constituted our study group. All had been admitted to the gynecology outpatient clinic between June 1, 2011, and January 1, 2012, for infertility treatment. Our control group consisted of 20 patients admitted for routine Pap smear control. They had no history of infertility and were not using contraceptives and they were actively attempting pregnancy. Midluteal progesterone measurement and pipelle endometrial biopsies were performed with both groups. For PCOS patients, metformin treatment was initiated right after the biopsy and CC was added in the second menstrual cycle. Pipelle endometrial biopsies were repeated. Histological dating and immunohistochemistry for glycodelin were performed by a single pathologist who was blinded to the patients clinical data. Result(s). The posttreatment ovulation rate in the study group was 93.3%. No pregnancies were achieved in either group when glycodelin expression was not present, even in the presence of ovulation. When glycodelin expression was high in PCOS group, the pregnancy rate was 60% and all pregnancies ended in live births. In weak expression group, however, three out of four pregnancies ended as early pregnancy losses. Conclusion(s). Endometrial glycodelin expression is an important predictor of pregnancy outcomes in both PCOS and fertile groups.
... lire plus sur ce sujet dans un nouvel onglet.... | | |  |
| Franks S: Polycystic Ovary Syndrome: A Changing Per spec tive. Clin Endocrinol 31:87-120, 1989 .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Chinese herbal medicine for subfertile women with polycystic ovarian syndrome .. cacher .... voir plus ..
Background Background Polycystic ovarian syndrome (PCOS) is one of the most common reproductive endocrinology abnormalities, and affects 5% to 10% of women of reproductive age. Western medicines, such as oral contraceptives, insulin sensitizers and laparoscopic ovarian drilling (LOD), have been used to treat PCOS. Recently, many studies have been published that consider Chinese herbal medicine (CHM) as an alternative treatment for women with PCOS. Objectives Objectives To assess the efficacy and safety of CHM for subfertile women with PCOS. Search methods Search methods We searched sources, including the following databases, from inception to 9 June 2016: the Cochrane Gynaecology and Fertility Group Specialized Register, the Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE, EMBASE, Allied and Complementary Medicine (AMED), PsycINFO, Chinese National Knowledge Infrastructure (CNKI), VIP, Wanfang and trial registries. In addition, we searched the reference lists of included trials and contacted experts in the field to locate trials. Selection criteria Selection criteria Randomized controlled trials (RCTs) that considered the use of CHM for the treatment of subfertile women with PCOS. Data collection and analysis Data collection and analysis Two review authors independently screened appropriate trials for inclusion, assessed the risk of bias in included studies and extracted data. We contacted primary study authors for additional information. We conducted meta-analyses. We used the odds ratios (ORs) to report dichotomous data, with 95% confidence intervals (CI). We assessed the quality of the evidence using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) methods. Main results Main results We included five RCTs with 414 participants. The comparisons in the included trials were as follows: CHM versus clomiphene, CHM plus clomiphene versus clomiphene (with or without ethinyloestradiol cyproterone acetate (CEA)), CHM plus follicle aspiration plus ovulation induction versus follicle aspiration plus ovulation induction alone, and CHM plus laparoscopic ovarian drilling (LOD) versus LOD alone. The overall quality of the evidence for most comparisons was very low. None of the included studies reported live birth rate, and only one study reported data on adverse events. When CHM was compared with clomiphene (with or without LOD in both arms), there was no evidence of a difference between the groups in pregnancy rates (odds ratio (OR) 1.98, 95% confidence interval (CI) 0.78 to 5.06; two studies, 90 participants, I² statistic = 0%, very low quality evidence). No study reported data on adverse events. When CHM plus clomiphene was compared with clomiphene (with or without CEA), there was low quality evidence of a higher pregnancy rate in the CHM plus clomiphene group (OR 2.62, 95% CI 1.65 to 4.14; three RCTs, 300 women, I² statistic = 0%,low quality evidence). No data were reported on adverse events. When CHM with follicle aspiration and ovulation induction was compared with follicle aspiration and ovulation induction alone, there was no evidence of a difference between the groups in pregnancy rates (OR 1.60, 95% CI 0.46 to 5.52; one study, 44 women, very low quality evidence), severe luteinized unruptured follicle syndrome (LUFS) (OR 0.60, 95% CI 0.06 to 6.14; one study, 44 women, very low quality evidence), ovarian hyperstimulation syndrome (OHSS) (OR 0.16, 95% CI 0.00 to 8.19; one study, 44 women, very low quality evidence) or multiple pregnancy (OR 0.60, 95% CI 0.06 to 6.14; one study, 44 women, very low quality evidence). When CHM with LOD was compared with LOD alone, there was no evidence of a difference between the groups in rates of pregnancy (OR 3.50, 95% CI 0.72 to 17.09; one study, 30 women, very low quality evidence), No data were reported on adverse events. There was no evidence of a difference between any of the comparison groups for any other outcomes. The quality of the evidence for all other comparisons and outcomes was very low. The main limitations in the evidence were failure to report live birth or adverse events, failure to describe study methods in adequate detail and imprecision due to very low event rates and wide CIs. Authors conclusions Authors conclusions There is insufficient evidence to support the use of CHM for women with PCOS and subfertility. No data are available on live birth, and there is no consistent evidence to indicate that CHM influences fertility outcomes. However there is very limited low quality evidence to suggest that the addition of CHM to clomiphene may improve pregnancy rates. There is insufficient evidence on adverse effects to indicate whether CHM is safe.
... lire plus sur ce sujet dans un nouvel onglet.... | | | 
|
| Lanzone A, Fulghesu AM, Andreani CL, et al: Insulin Se cre tion in Polycystic Ovarian Disease: Effect of
Ovarian Sup pres sion by GnRH Agonist. Hum Reprod 5:143-149, 1990. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Insulin-sensitising drugs (metformin, rosiglitazone, pioglitazone, D-chiro-inositol) for women with polycystic ovary syndrome, oligo amenorrhoea and subfertility .. cacher .... voir plus ..
Background Background Polycystic ovary syndrome (PCOS) is characterised by infrequent or absent ovulation (anovulation), high levels of male hormones (hyperandrogenaemia) and high levels of insulin (hyperinsulinaemia secondary to increased insulin resistance). Hyperinsulinaemia is associated with an increase in cardiovascular risk and the development of diabetes mellitus. Insulin-sensitising agents such as metformin may be effective in treating the features of PCOS, including anovulation. Objectives Objectives To assess the effectiveness of insulin-sensitising drugs in improving reproductive outcomes and metabolic parameters for women with PCOS. Search methods Search methods We searched the Cochrane Menstrual Disorders and Subfertility Group Trials Register (October 2011), the Cochrane Central Register of Controlled Trials (CENTRAL) (The Cochrane Library, 3rd Quarter 2011), CINAHL (October 2011), MEDLINE (January 1966 to October 2011), and EMBASE (January 1985 to October 2011). Selection criteria Selection criteria Randomised controlled trials of insulin sensitising drugs compared with either placebo, no treatment, or an ovulation induction agent for women with PCOS, menstrual disturbance and subfertility. Data collection and analysis Data collection and analysis Two review authors independently assessed studies for inclusion and trial quality, and extracted data. Main results Main results Forty-four trials (3992 women) were included for analysis, 38 of them using metformin and involving 3495 women. There was no evidence that metformin improved live birth rates, whether it was used alone (pooled OR 1.80, 95% CI 0.52 to 6.16, 3 trials, 115 women) or in combination with clomiphene (pooled OR 1.16, 95% CI 0.85 to 1.56, 7 trials, 907 women). However, clinical pregnancy rates were improved for metformin versus placebo (pooled OR 2.31, 95% CI 1.52 to 3.51, 8 trials, 707 women) and for metformin and clomiphene versus clomiphene alone (pooled OR 1.51, 95% CI 1.17 to 1.96, 11 trials, 1208 women). In the studies that compared metformin and clomiphene alone, there was evidence of an improved live birth rate (pooled OR 0.3, 95% CI 0.17 to 0.52, 2 trials, 500 women) and clinical pregnancy rate (pooled OR 0.34, 95% 0.21 to 0.55, 2 trials, 500 women) in the group of obese women who took clomiphene. Metformin was also associated with a significantly higher incidence of gastrointestinal disturbances than placebo (pooled OR 4.27, 95% CI 2.4 to 7.59, 5 trials, 318 women) but no serious adverse effects were reported. Authors conclusions Authors conclusions In agreement with the previous review, metformin was associated with improved clinical pregnancy but there was no evidence that metformin improves live birth rates whether it is used alone or in combination with clomiphene, or when compared with clomiphene. Therefore, the role of metformin in improving reproductive outcomes in women with PCOS appears to be limited.
... lire plus sur ce sujet dans un nouvel onglet.... | | |  |
| Farah L, Azziz R: Polycystic Ovary Syndrome. The Female Patient. 24: 79-84, 1999. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Tamoxifen: an alternative approach in clomiphene resistant polycystic ovarian syndrome patients .. cacher .... voir plus ..
Twenty patients with ultrasound diagnosed polycystic ovaries and high luteinizing hormone levels who failed to respond to clomiphene citrate were given tamoxifen to induce ovulation. Tamoxifen 20 mg daily (group 1, n = 10) and 40 mg daily (group 2, n = 10) was given for 5 consecutive days from the third to seventh day of the cycle. Treatment was monitored by serial ultrasound scans and assessment of serum estradiol. Human chorionic gonadotrophin (hCG) was administered when at least one follicle was > 16mm and serum estradiol level was > 300 pg/ml per follicle. Ovulation was confirmed with detection of the follicular rupture ultrasonographically 2 days after hCG and midluteal progesterone levels. The ovulation rate achieved in group 2 patients was significantly higher (p = 0.01) than group 1. Three pregnancies were achieved in group 2 patients while there was no pregnancy in group 1. The only side effect was ovarian cyst formation in one patient in group 1. As a result, tamoxifen might be a good choice for clomiphene resistant patients prior to treatment with surgery or hMG.
... lire plus sur ce sujet dans un nouvel onglet.... | | |  |
| Effects of Metformin on Spontaneous and Clomiphene-Induced Ovulation in the Polycystic Ovary Syndrome .. cacher .... voir plus ..
Polycystic ovary syndrome, which affects approximately 6 percent of women of reproductive age and is characterized by chronic anovulation and hyperandrogenism,1 is the most common cause of infertility in women in the United States. Insulin resistance with compensatory hyperinsulinemia is a prominent feature of the syndrome2–5 and appears to have a pathophysiologic role in the hyperandrogenism of the disorder. Ovarian androgen production and serum free testosterone concentrations decrease in women with polycystic ovary syndrome when insulin secretion is reduced by drugs such as diazoxide,6 metformin,7–10 and troglitazone.11,12 However, whether such therapy improves ovulatory function is not known. . . .
... lire plus sur ce sujet dans un nouvel onglet.... | | | 
|
| Farah L, Lazenby AJ, Boots LR, et al: Prevalence of Poly cys tic Ovarian Syndrome in Women Seeking Treatment
from Community Electrologists. J Repro Med 44:870-874, 1999. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| L'endométriose est associée à une incidence accrue de SPOK chez ces patientes qui présentent une infertilité.
Mukherjee, T, Addulla M, Copperman TK: Increased In ci dence of Endometriosis Co ex ist ing with Poly cys tic
Ovary Syndrome in Infertile Patients. Poster presentation 1998 54th Annual Meeting ASRM San Francisco,
CA. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Extended-Release Metformin Does Not Reduce the Clomiphene Citrate Dose Required to Induce Ovulation in Polycystic Ovary Syndrome .. cacher .... voir plus ..
Context: When used for ovulation induction, higher doses of clomiphene may lead to antiestrogenic side effects that reduce fecundity. It has been suggested that metformin in combination with clomiphene can restore ovulation to some clomiphene-resistant anovulators with polycystic ovary syndrome (PCOS)., Objective: Our objective was to determine if cotreatment with extended-release metformin (metformin XR) can lower the threshold dose of clomiphene needed to induce ovulation in women with PCOS., Design: A secondary analysis of data from the National Institute of Child Health and Human Development Cooperative Multicenter Reproductive Medicine Network prospective, double-blind, placebo-controlled multicenter clinical trial, Pregnancy in Polycystic Ovary Syndrome, was performed., Setting: Study volunteers at multiple academic medical centers were included., Participants: Women with PCOS and elevated serum testosterone who were randomized to clomiphene alone or with metformin (n = 209 in each group) were included in the study., Interventions: Clomiphene citrate, 50 mg daily for 5 d, was increased to 100 and 150 mg in subsequent cycles if ovulation was not achieved; half also received metformin XR, 1000 mg twice daily. Treatment was for up to 30 wk or six cycles, or until first pregnancy., Main Outcome Measures: Ovulation was confirmed by a serum progesterone more than or equal to 5 ng/ml, drawn prospectively every 1–2 wk., Results: The overall prevalence of at least one ovulation after clomiphene was 75 and 83% (P = 0.04) for the clomiphene-only and clomiphene plus metformin groups, respectively. Using available data from 314 ovulators, the frequency distribution of the lowest clomiphene dose (50, 100, or 150 mg daily) resulting in ovulation was indistinguishable between the two treatment groups., Conclusion: Metformin XR does not reduce the lowest dose of clomiphene that induces ovulation in women with PCOS., In anovulatory women with polycystic ovary syndrome seeking fertility, metformin does not decrease the clomiphene dose needed to induce ovulation.
... lire plus sur ce sujet dans un nouvel onglet.... | | |  |
| Dahlgren E, Johansson S, Lindstedt G, et al: Women with Polycystic Ovary Syndrome Wedge Resected in
1956 to 1965: A Long-Ter .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Long versus short course treatment with metformin and clomiphene citrate for ovulation induction in women with PCOS (Review) .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | | 
|
| Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Work shop Group: Revised 2003 Consensus on
Di ag nos tic Criteria and Long-Term Health Risks Related to Polycystic Ovary Syndrome. Fertil Steril 81:19-
25, 2004 .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Vers 40 ans, 40% des femmes avec un SPOK développent du diabète de type II.
Thacher SS: PCOS: The Hidden Epidemic. Perspectives Press, Indianapolis, IN, 2001. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | Source: Niveau de preuve non connu |
| Clomiphene citrate or letrozole as first-line ovulation induction drug in infertile PCOS women: A prospective randomized trial .. cacher .... voir plus ..
OBJECTIVE: To compare Letrozole (5 mg) and clomiphene citrate (100 mg) as first line ovulation induction drug in infertile PCOS women. STUDY DESIGN: Prospective Randomised trial. SETTING: A Tertiary level infertility centre. Patients: 103 infertile PCOS women INTERVENTION(S): Treatment naïve infertile PCOS women were randomised to treatment with 5 mg letrozole (51 patients) or 100 mg clomiphene citrate (52 patients) daily starting day 2 to day 6 of menstrual cycle. Timed intercourse or Intra Uterine Insemination (IUI) was advised 24 to 36 hours after Human Chorionic Gonadotropin (HCG) injection. MAIN OUTCOME MEASURES: Ovulation rate, mono or multi follicular rate, days to ovulation, endometrial thickness, serum progesterone, serum estrogen, pregnancy rate, miscarriage rate. RESULTS: The mean age, Body Mass Index (BMI), duration of infertility in both Clomiphene Citrate (CC) and Letrozole groups were similar.Ovulation rate was 73.08% in letrozole group and 60.78% in CC, which was not statistically significant (P=0.398). There was no statistically significant difference between Endometrial thickness (CC 7.61 ±1.96, Let 7.65 ± 2.10), Sr E2 on day of HCG (CC 178.3 ± 94.15, Let 162.09 ± 73.24), Days to ovulation (CC 14.2 ± 3.41; Let 13.13 ± 2.99) and Sr P4 on D21 (CC 10.58 ± 6.65; Let 11.86 ± 6.51). Monofolliculo genesis (CC 54.84, Let 79.49 %, P=0.027) and Pregnancy rate (CC 7.84%, Let 21.56% P=0.0125) were statistically significantly higher in letrozole group. CONCLUSION: Our study shows that letrozole has excellent pregnancy rates compared to clomiphene citrate. Letrozole should be considered at par with clomiphene citrate as first line drug for ovulation induction in infertile PCOS women.
... lire plus sur ce sujet dans un nouvel onglet.... | | |  |
| Gibson M: Reproductive Health and Polycystic Ovary Syn drome. Am J Med 98:675-755, 1995 .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Evaluating the equivalence of clomiphene citrate with and without metformin in ovulation induction in PCOS patients .. cacher .... voir plus ..
Objective To evaluate the benefit of Metfomin added to Clomiphene Citrate in a primary ovulation induction protocol in PCOS patients Design Prospective randomised controlled study Setting Tygerberg Academic Hospital, Stellenbosch University and the Institute of Reproductive Medicine at Vincent Pallotti Hospital, Cape Town Patients 107 patients presenting with PCOS Study Group A was pre-treated with metformin 850 mg twice a day for at least 6 weeks before clomiphene was added and the metformin was used throughout the study period. Group B received clomiphene without pre-treatment with metformin. In both groups clomiphene was given at a starting dose of 50 mg day 4–8 and increase with increments of 50 mg to a maximum of 150 mg if no response was achieved. Results The ovulation rate achieved in women in the M+C/C arm was 34/52 (65.4%) compared to 36/55 (65.5%) in the C/C arm. The treatment effect ((M+C/C) – C/C) is 0% with 95% confidence interval of −18.1% to 18%. The per protocol ovulation results were 34/42 (81%) in the M+C/C arm compared to 36/48 (75%) in the C/C arm. The ovulation rate difference was 6% with 95% confidence interval −11% to 22%. In a comparison of successful ovulating versus non-ovulating women from the trial the following were significant baseline determinants: lower median weight in the ovulating group (77 kg versus 86 kg, p = .021), lower median bmi (29.0 versus 32.9, p = .009), lower median DHEAS at baseline (4.6 compared to 7.0, p = .049), lower median 17OH-progesterone (2.2 versus 4.6, p = .027) and higher baseline median SHBG ( 37.8 compared to 28.5, p = .036). Conclusion Although identical ovulation rates were observed in both arms equivalence could not be concluded with respect to the specified criteria.
... lire plus sur ce sujet dans un nouvel onglet.... | | | 
|
| Kurabayashi T, Kase H, Suzuki M, et al: Endometrial Ab nor mal i ties in Infertile Women. J Reprod Med.
48:455-459, 2003. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Role of Anti-Müllerian Hormone in pathophysiology, diagnosis and treatment of Polycystic Ovary Syndrome: a review .. cacher .... voir plus ..
Polycystic ovary syndrome (PCOS) is the most common cause of chronic anovulation and hyperandrogenism in young women. Excessive ovarian production of Anti-Müllerian Hormone, secreted by growing follicles in excess, is now considered as an important feature of PCOS. The aim of this review is first to update the current knowledge about the role of AMH in the pathophysiology of PCOS. Then, this review will discuss the improvement that serum AMH assay brings in the diagnosis of PCOS. Last, this review will explain the utility of serum AMH assay in the management of infertility in women with PCOS and its utility as a marker of treatment efficiency on PCOS symptoms. It must be emphasized however that the lack of an international standard for the serum AMH assay, mainly because of technical issues, makes it difficult to define consensual thresholds, and thus impairs the widespread use of this new ovarian marker. Hopefully, this should soon improve.
... lire plus sur ce sujet dans un nouvel onglet.... | | |  |
| Rutanen EM, Stenman S, Blum W, et al: Relationship be tween Carbohydrate Metabolism and Serum
Insulin-like Growth Factor System in Postmenopausal women: Com par i son of Endometrial Cancer Patients
with Healthy Con trols. J Clin Endocrinol Metab 77:199-204, 1993. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Nagaman N, Hannigan EV, Van Dinh T, et al: Hyperinsulinemia and Stromal Lutein iza tion of the Ovaries
in Postmenopausal Women with Endometrial Cancer. J Clin Endocrinol Metab 67:144-148, 1988. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Kazeer RR: Insulin Resistance, Insulin-like Growth Factor-I and Breast Cancer: An Hy poth e sis. Int J Cancer
62:403-406, 1995. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Coulam CB, Annegers JF, Kranz JS: Chronic Anovulation Syndrome and Associated Neo pla sia. Obstet
Gynecol 61:403-407, 1983. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Coulam CB, Annegers JF, Kranz JS: Chronic Anovulation Syndrome and Associated Neo pla sia. Obstet
Gynecol 61:403-407, 1983. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Kaaks R: Nutrition, Hormones and Breast Cancer: Is Insulin the Missing Link? Cancer Causes Control.
7:605-625, 1996. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Secreto G, Zumoff B: Abnormal Production of Androgens in Women with Breast Cancer. Anticancer Res
14:2113-2117, 1994. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Les femmes atteintes de SOPK ont un risque d'infarctus du myocarde et de cardiopathies augmenté d'au moins sept fois par rapport aux autres femmes.
Dahlgren E, Janson PO, Johansson S, et al: Polycystic Ova ry Syndrome and Risk for Myocardial In f arc tion.
Acta Obstet Gyncol Scand. 71:599-604, 1992. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Solomon CG: The Epidemiology of Polycystic Ovary Syn drome. Prevalence and As so ci at ed Disease Risks.
Endocrinol Metab Clin North Am 28:247-263, 1999. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Yarali H, Yildirir A, Aybar F, et al: Diastolic Dysfunction and Increased Serum Ho mocys teine Concentrations
May Con trib ute to Increased Cardiovascular Risks in Patients with Polycystic Ovary Syndrome. Fertil Steril
76:511-516, 2001. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Harlow BL, Signorello LB, Hall JE, et al: Reproductive Cor re lates of Chronic Fatigue Syndrome. Am J Med.
105(3A):94S-99S, 1998. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Hansen KA: Hirsutism: Options for Treating the Whole Patient. The Female Patient 23:31-38, 1998. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Schleicher S: Hirsutism: Looking Below the Surface. The Female Patient 25:38-44, 2000. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Mastorakos G, Koliopoulos C, Creatsas G: Androgen and Lipid Profi les in Adolescents with Polycystic Ovary
Syn drome Who were Treated with Two Forms of Combined Oral Con tra cep tives. Fertil Steril 77:919-927,
2002. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Milewicz A, Silber D, Kirschner MA: Therapeutic Effects of Spironolactone in Polycystic Ovary Syndrome.
Obstet Gynecol 61:429-432, 1983. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Blum I, Kaufman H, Marilus R, et al: Successful Treatment of Polycystic Ovary Syndrome with Spironolactone
or Bromocriptine. Obstet Gynecol 57:661-665, 1981 .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Serafi ni P, Lobo RA: The Effects of Spironolactone on Adrenal Steroidogenesis in Hirsute Women. Fertil
Steril 44:595-599, 1985. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Unluhizarci K, Everest H, Bayram F, et al: Comparison of Spironolactone and Spirono lac tone plus Finasteride
in the Treatment of Hirsutism. Fertil Steril 78:1331-1332, 2002. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Dunaif A, Thomas A: Current Concepts in the Polycystic Ovary Syndrome. Annu Rev Med 52:401-419,
2001. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Yildirim B, Sabir N, Kaleli B: Relation of Intra-Abdominal Fat Distribution to Metabolic Disorders in
Non-obese Pa tients with Polycystic Ovary Syndrome. Fertil Steril 79:1358-1364, 2003. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Nestler JE: Should Patients with Polycystic Ovarian Syn drome be Treated with Metformin? An Enthusiastic
En dorse ment. Hum Reprod 17:1950-1953, 2002. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Marca A, Morgante G, Palumbo M, et al: Insulin-Lowering Treatment Reduces Aromatase Activity in Response
to Fol li cle-Stimulating Hormone in Women with Polycystic Ova ry Syndrome. Fertil Steril 78:1234-1239,
2002. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Morin-Papunen LC, Koivunen RM, Ruokonen A, et al: Metformin Therapy Improves the Menstrual Pattern
with Minimal Endocrine and Metabolic Effects in Women with Poly cys tic Ovary Syndrome. Fertil Steril
69:691-696, 1998. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Velzquez E, Acosta A, Mendoza SG: Menstrual Cyclicity After Metformin Therapy and Polycystic Ovary
Syn drome. Obstet Gynecol 90:329-395, 1997. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Vandermolen DT, Ratts VS, Evans WS, et al: Metformin Increases the Ovulatory Rate and Pregnancy Rate
from Clo mi phene Citrate in Patients with Polycystic Ovary Syn drome Who are Resistant to Clomiphene
Citrate Alone. Fertil Steril 75:310-315, 2001. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Costello MF, Eden JA: A Systematic Review of the Re pro duc tive System Effects of Metformin in Patients
with Poly cys tic Ovary Syndrome. Fertil Steril 79:1-13, 2003. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Lord JM, Flight IHK, Norman RJ: Metformin in Polycystic Ovary Syndrome: Systematic Review and
Meta-Analysis. Br Med J 327:1-6, 2003. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Lanzone A, Caruso A, Di Simone N, et al: Polycystic Ovary Disease, A Risk Factor for Gestational Diabetes?
J Reprod Med 40:312-316, 1995 .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Anttila L, Karjala K, Pentilla T-A, et al: Polycystic Ovaries in Women with Gestational Diabetes. Obstet
Gynecol 92:13-16, 1998. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Glueck CJ, Wang P, Kobayashi S, et al: Metformin Therapy Throughout Pregnancy Reduces the Development
of Ges ta tion al Diabetes in Women with Polycystic Ovary Syn drome. Fertil Steril 77:520-525, 2002. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Craig LB, Ke RW, Kutteh WH: Increased Prevalence of Insulin Resistance in Women with a History of
Recurrent Pregnancy Loss. Fertil Steril 78:487-490, 2002. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Glueck CJ, Phillips H, Cameron D, et. al: Continuing Metformin Throughout Pregnancy in Women with
Polycystic Ovary Syndrome Appears to Safely Reduce First-Trimester Spontaneous Abortion: A Pilot Study.
Fertil Steril 75:46-52, 2001. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Kaaks R: Nutrition, Hormones and Breast Cancer: Is Insulin the Missing Link? Cancer Causes Control.
7:605-625, 1996. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Legro RS, Urbanek M, Kunselman AR, et al: Self-Selected Women with Poly cys tic Ovary Syndrome are
Re pro duc tive ly and Metabolically Abnormal and Under Treated. Fertil Steril 78:51-57, 2002. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Mukherjee, T, Addulla M, Copperman TK: Increased In ci dence of Endometriosis Co ex ist ing with Poly cys tic
Ovary Syndrome in Infertile Patients. Poster presentation 1998 54th Annual Meeting ASRM San Francisco,
CA. .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Secreto G, Zumoff B: Abnormal Production of Androgens in Women with Breast Cancer. Anticancer Res
14:2113-2117, 1994 .. cacher .... voir plus ..
... lire plus sur ce sujet dans un nouvel onglet.... | | |
| Anomalie ou Maladie concernée : | | |
| Syndrome des ovaires polykystiques .. cacher .... voir plus ..
Le syndrome des ovaires polykystiques (SOPK) est un trouble endocrinien (hormonal). Le plus souvent, les symptômes apparaissent pour la première fois à l'adolescence, lors des premières menstruations. Cependant, certaines femmes ne développent pas de symptômes avant le milieu de la vingtaine.
Aucune femme n'a exactement les mêmes symptômes. Les caractéristiques suivantes sont très souvent associées au SOPK, mais toutes ne sont pas visibles chez chaque femme:
Hirsutisme (croissance excessive des cheveux sur le visage, poitrine, abdomen, etc.)
Perte de cheveux (alopécie androgénique, dans un schéma classique de «calvitie masculine»)
Acné
Ovaires polykystiques
Obésité
Infertilité ou fertilité réduite
En outre, les femmes souffrant de SOPK semblent être exposées à un risque accru de développer les problèmes de santé suivants au cours de leur vie:
Résistance à l'insuline
Diabète
Anomalies lipidiques
Apnée obstructive du sommeil
Maladie cardiovasculaire
Cancer de l'endomètre
Étant donné qu'il existe une telle variabilité dans la présentation clinique du SOPK, il n'existe pas d'accord universel parmi les professionnels de la santé sur la manière de mieux définir le syndrome. Ce qui est clair, cependant, c'est que les femmes souffrant de SOPK n'ovulent pas de manière prévisible et produisent également des quantités excessives d'androgènes (testostérone libre et totale, DHEAS).
Les ovaires polykystiques ne sont pas présents chez toutes les femmes diagnostiquées avec SOPK. En outre, de nombreuses femmes ayant des cycles réguliers et un niveau normal de testostérone ont des ovaires kystiques.
Il est important de faire la distinction entre les ovaires polykystiques et le syndrome des ovaires polykystiques.
Les ovaires polykystiques sont souvent, mais pas toujours, observés chez les femmes atteintes de SOPK.
Et environ 20 pour cent des femmes sans anomalies menstruelles ou hormonales ont des ovaires polykystiques.
Le syndrome est donc défini par les anomalies menstruelles et hormonales avec ou sans ovaires polykystiques.
... lire plus sur ce sujet dans un nouvel onglet.... | 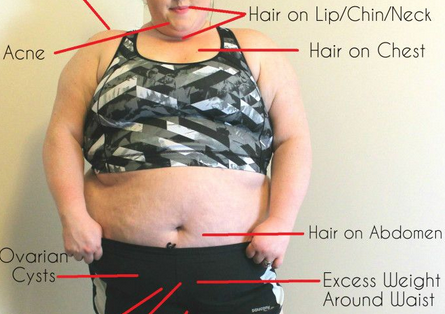 | |
 Médecine par les plantes, la nutrition et l'activité, mode de vie, acupuncture
Médecine par les plantes, la nutrition et l'activité, mode de vie, acupuncture Méthodes naturelles de régulation des naissances
Méthodes naturelles de régulation des naissances Restauration de la fertilité, anomalies du cycle, etc..
Restauration de la fertilité, anomalies du cycle, etc.. Traitement des kystes ovariens récidivants
Traitement des kystes ovariens récidivants